
03

Technical Information01.
Crystallization Containers and Methods
Technical Information02.
Protein Characterization as Quality Control
The JCB-SGT container is the standard container used for protein crystallization in JAXA's PCG (protein crystal growth) experiments.
A JCB-SGT container is made of polyethylene terephthalate (PET) sheets and has three thin cylindrical cells. Each JCB-SGT container can hold three different conditions. The container has a very low gas permeability and virtually no solution volume change.
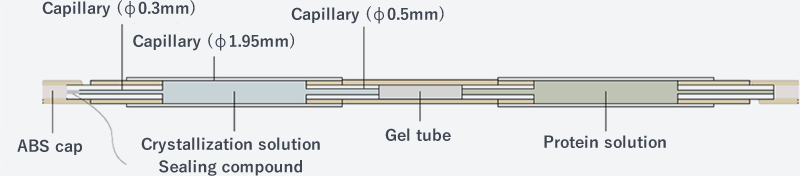
Each cell is designed to contain up to two capillaries containing crystallization solution and protein solution. Setting up the containers is carried out on Earth and transported to the International Space Station (ISS).
The standard capillary has an inner diameter of 0.5 mm. Capillaries of inner diameters ranging from 0.3 to 3.0 mm can be chosen. In addition to the JCB-SGT DX for creating deoxidized environment, several other crystallization methods are available as options.
*: Abbreviated name for JAXA Crystallization Box - Sealbag Gel Tube.

This crystallization container is originally developed for the U.S. flights. It is made of PET sheets composed of two separate cells to hold two different solutions. A crew member squeezes it with fingers to brake the seal so that the two solutions are mixed and trigger crystallization on orbit.
Usually, one cell contains a capillary filled with a protein solution and a stabilizing buffer and the other cell is filled with crystallization solution.
A desired amount of solution can be chosen for each of the cells.


Counter diffusion method is the standard method for the JAXA PCG experiments at present. Other crystallization methods can be used depending on the conditions of samples and the objectives of the experiment.
Some typical crystallization methods usable for the JAXA PCG are described below.
The counter diffusion (CD) method is categorized as a solution-solution diffusion technique. The basic unit used for the current JAXA PCG experiments consists of a silicone tube filled with agarose gel (gel tube) attached to a glass capillary, which is filled with a protein solution.
A crystallization solution is diffused through the gel tube into the capillary, while protein solution is diffused through the gel tube from the capillary (the diffusion speeds of solute molecules depend on their molecular weights). As a result of these diffusions in opposite directions, a wide range of precipitant concentration can be explored at one time.
The CD method, which prevents protein from being condensed during crystallization, enables crystal growth in a gentler manner than vapor diffusion methods. In several cases, the quality of crystals was improved by changing the method from the vapor diffusion method to the CD method.

Mechanism of crystallization by the CD method

Configuration of CD method
The dialysis method is a technique for diffusing a crystallization solution through a dialysis membrane into a protein solution. Generally, a small container called "dialysis button" is used in the method. JAXA PCG uses a dialysis membrane inserted between capillary and gel tube instead of a dialysis button. Both dialysis methods are based on the same crystallization principle. It is expected that the concentration of the solute slowly changes as it has a longer diffusion path than a dialysis button.
The advantage of the dialysis method is to prevent the protein in the capillary from diffusing out, particularly suitable for producing large-sized crystals for neutron diffraction experiment.

Configuration of dialysis method
The osmosis tube (OT) method is a crystallization technique using a moisture permeable polymer membrane. There are two types: involving or not involving a glass capillary. In both types, the sample portion is filled with a mixture of protein solution and crystallization solution. Only water molecules can pass through the polymer membrane under the difference in osmotic pressure to the external solution. The OT method is a technique that is expected to similar results as a vapor diffusion method.
Each of the preceding methods uses an excessively larger amount of crystallization solution than the amount of solution in the sample portion (the standard amount of solution is several μL and that of the crystallization solution is approximately 1 mL). Therefore, it is estimated that once a sufficiently long diffusion time elapses, the composition of the material in the sample portion will become roughly the same as that of the crystallization solution except for protein concentration. This configuration usually causes no serious problem. But if the amount of the crystallization solution should be minimized as far as the things permit, for example when a precious substrate or inhibitor is used, the diffusion couple method is valid. With this method, the capillary filled with crystallization solution is connected with the sample portion by means of a gel tube.
See here for the filling of the gel tube method.
Protocol for filling with the gel-tube methodFor more detailed description, click below or write to: Z-crystal@ml.jaxa.jp
JCB-SGT Crystallization Devices Applicable to PCG Experiments and their Crystallization Conditions